A lot of formulators first ask what does hemp derived mean when a supplier sends over a distillate spec sheet that looks clean, legal, and ready to run. The label sounds simple. The operational reality usually isn't.
If you're building vape cartridges, concentrates, or other inhalable products, “hemp-derived” affects far more than a compliance checkbox. It affects how the cannabinoid was sourced, whether it was likely converted, what contaminants you need to screen for, how it behaves with your terpene system, and whether your finished product stays consistent batch after batch.
That matters most when you're buying inputs, not when you're writing packaging copy. A compliant input can still be a poor formulation input. A potent distillate can still carry process risk. And a technically legal ingredient can still wreck flavor accuracy if the purification work wasn't done properly.
Navigating the World of Hemp-Derived Ingredients
A common scenario goes like this. You're evaluating a new cannabinoid supplier for a cartridge line. They offer “hemp-derived Delta-9 distillate,” the price is competitive, and the potency looks strong on the front page of the COA.
That still leaves the fundamental questions unanswered.
Did the material come from naturally occurring THC in compliant hemp, or was it converted from CBD? Was the purification work thorough enough for inhalation use? Does the oil carry residual process notes that will push your terpene blend off target? If you're trying to replicate a clean fruit-forward or gas-forward profile, those details matter more than the sales sheet.
What the label tells you and what it doesn't
“Hemp-derived” does tell you something important. It tells you the ingredient is represented as coming from hemp rather than marijuana under the federal classification that governs hemp inputs.
It does not tell you:
- How the cannabinoid was made: Native extraction and post-extraction conversion are very different sourcing paths.
- How clean the distillate is: Purity on paper isn't the same as a complete contaminant screen.
- How the oil will taste in a formula: Some lots look acceptable analytically but still carry reactive or harsh background notes.
- Whether the supplier understands formulation use: A vendor selling bulk cannabinoids may not understand cartridge performance, terpene retention, or flavor replication.
Practical rule: Treat “hemp-derived” as a legal sourcing descriptor, not a quality descriptor.
In practice, new formulators get into trouble when they assume compliance language equals formulation readiness. It doesn't. A distillate that's fine for one product type may be a poor fit for a high-terpene vape application.
The formulator's lens
From a product development standpoint, the useful question isn't just what does hemp derived mean in law. It's what that term implies for your workflow.
For most buyers, it should trigger a review of four things:
- Legal status of the starting material
- Probability of CBD conversion
- Depth of analytical testing
- Compatibility with your terpene target
Those four factors usually tell you whether you're looking at a workable input or a future customer complaint.
The Legal Line The 2018 Farm Bill and 0.3% THC Rule
Hemp-derived is a legal classification first. The term comes from the Agricultural Improvement Act of 2018, commonly called the 2018 Farm Bill. Under that law, cannabis plants with 0.3% or less Delta-9 THC by dry weight are classified as hemp, while material above that threshold falls on the marijuana side of the federal line, as explained in Longleaf Pro's breakdown of the 2018 Farm Bill definition.

That legal distinction changed the supply chain. The same source notes that the legislation legalized cultivation and sale of hemp-derived products and that U.S. hemp acreage surged over 400% in the first year post-Bill. For formulators, that expansion is why hemp-derived cannabinoids became so widely available in the first place.
What the dry weight rule means in practice
The phrase “by dry weight” is where a lot of confusion starts. In raw plant material, the concept is straightforward. In processed goods, it gets more complicated because the total product weight affects whether a given amount of Delta-9 fits under the federal threshold.
For a formulator, the key point is simple. You don't evaluate legality by marketing language or by cannabinoid family alone. You evaluate it by whether the source material and resulting product fit the legal definition tied to Delta-9 THC concentration on a dry-weight basis.
That means your documentation has to connect:
- The source biomass
- The extraction or conversion pathway
- The cannabinoid profile of the finished ingredient
- The final product format
Why this matters before you formulate
Teams often focus on flavor development first and compliance second. That's backwards. If the legal classification of your input is shaky, every downstream decision gets harder, including shipping, manufacturing, insurance, and retail placement.
A practical purchasing habit is to separate three documents in your review process:
| Document | What it should confirm | Why it matters |
|---|---|---|
| Biomass or source record | Hemp origin | Supports the legal basis of the ingredient |
| COA | Potency and safety data | Confirms what's actually in the material |
| Product spec sheet | Handling and use details | Helps determine formulation fit |
One more point. Federal classification doesn't answer every operational question you may have around testing, handling, or end use. It only gives you the legal line that starts the conversation.
If your team also fields common compliance questions from retailers or customers, resources like this article on whether CBD shows up in a blood test can help clarify adjacent misunderstandings without changing your internal QC standards.
The most expensive mistake is building a polished product around an ingredient that wasn't properly vetted at the legal definition stage.
Hemp-Derived vs Cannabis-Derived A Chemical Reality Check
A lot of buyers still talk about hemp-derived THC and cannabis-derived THC as if they're different active molecules. They aren't. The important distinction is source and process, not the identity of the finished Delta-9 THC molecule.
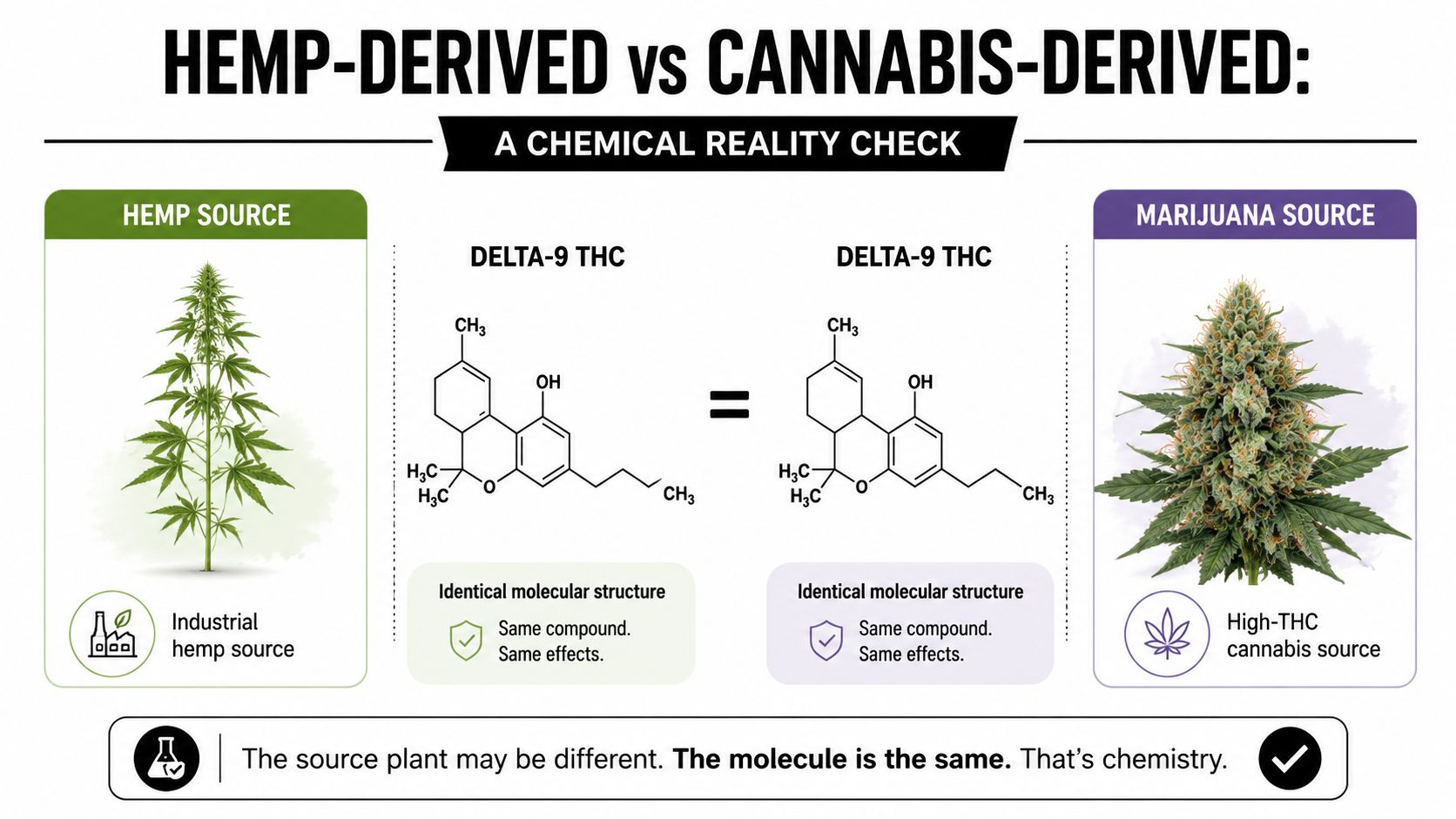
According to H Hemp's explanation of hemp-derived cannabinoids, Δ9-THC molecules from hemp and marijuana are bioidentical, but hemp sourcing usually requires chemical conversion because hemp naturally carries very low native Δ9-THC. The same source explains the breeding difference clearly: hemp tends to show CBD:THC ratios greater than 100:1, while marijuana is described at 1:20.
Side by side reality for formulators
That creates two very different production paths.
| Factor | Hemp source | Marijuana source |
|---|---|---|
| Primary cannabinoid abundance | Usually CBD-rich | Usually THC-rich |
| Typical path to commercial Delta-9 | Often conversion from CBD | Direct extraction is more straightforward |
| Core buying question | How clean was the conversion and cleanup? | How clean was the extraction and refinement? |
| Legal framing | Governed by hemp definition | Separate legal category |
From a chemistry standpoint, once you have purified Delta-9 THC, the molecule doesn't carry a memory of where it came from. It won't behave differently in a formulation because one batch started from hemp and another started from marijuana.
What changes is the risk profile of the manufacturing journey.
Why the process matters more than the label
If you formulate strain-inspired products, this distinction becomes practical fast. A terpene system built to replicate a sharp citrus top note, a floral middle, and a dense resinous finish needs a neutral enough cannabinoid base to let those layers show up as intended.
Converted hemp-derived inputs can work well. Poorly cleaned ones usually don't.
Common differences a formulator notices aren't molecular effects. They're process artifacts:
- Background odor: Slightly sour, solvent-like, or chemically flat notes
- Color inconsistency: A clue that purification or post-processing varied
- Flavor suppression: Top notes disappear sooner than expected
- Harshness in vapor: Not always a terpene issue. Sometimes the base oil is the problem
If you're comparing botanical and cannabis-style aroma systems, this guide to CDT vs BDT differences in formulation is useful because it frames sensory choices around composition rather than hype.
Buyers who focus only on potency usually miss the bigger formulation question. Can this oil carry the profile you want without fighting it?
What works and what doesn't
What works is a neutral, well-refined cannabinoid input paired with a terpene profile designed for the finished hardware and target flavor structure.
What doesn't work is trying to fix a contaminated or reactive base oil with more terpenes. More flavor doesn't solve bad chemistry. It usually amplifies instability, increases harshness, and muddies the profile.
The Formulator's Guide to Converted Cannabinoids
Most commercial hemp-derived Delta-9 on the market doesn't come from naturally abundant Delta-9 in hemp flower. It comes from CBD conversion. That's the part many supplier decks soften, even though it's the first thing a formulator should understand before buying.

Redeem Rx explains the process in practical terms. Most hemp-derived delta-9 THC is created by chemically converting abundant CBD. The same source states that acid-catalyzed cyclization can convert over 70% of CBD to THC, followed by chromatography that can isolate THC to greater than 95% purity.
Why conversion is necessary
Hemp is a legal source of cannabinoids, but it isn't naturally a rich source of Delta-9 THC. That's why the industry leans on CBD as the starting feedstock. CBD is abundant enough in hemp to make the economics workable, and the chemistry allows manufacturers to rearrange that molecule into THC through controlled reactions.
At a high level, the path looks like this:
- Extract CBD-rich oil from hemp
- Purify the CBD stream
- Use acid-catalyzed conversion to produce THC isomers
- Refine the reaction mixture
- Isolate and test the target cannabinoid
That description sounds tidy. Real production often isn't.
Where quality problems start
Conversion chemistry introduces a second layer of formulation risk because your input isn't just extracted. It's extracted, reacted, and then cleaned up. Every one of those stages can leave a trace behind if the process control isn't strong.
The common blind spots are straightforward:
- Residual solvents from extraction or reaction cleanup
- Acid residues if neutralization and washing weren't handled well
- Unwanted byproducts from side reactions
- Incomplete conversion that leaves a more complex cannabinoid mix than the label suggests
- Sensory carryover that doesn't violate a potency spec but still damages flavor
A surprising number of buying mistakes happen because the purchaser sees one strong potency number and assumes the material is formulation-ready. It may only be potency-ready.
Bench insight: If a converted distillate has even a faint off-note before terpenes are added, don't assume the blend will cover it. Heat and aerosolization usually make low-level defects easier to detect, not harder.
This walkthrough is worth watching if your team needs a visual primer on conversion concepts and cannabinoid handling in production.
What to ask a supplier before approving a lot
You don't need a supplier to reveal proprietary process details. You do need enough information to judge whether the ingredient is fit for your product.
Ask questions like these:
Was the Delta-9 directly extracted or converted from CBD?
If it was converted, the rest of your review gets stricter.Which analytical panels are included beyond potency?
A potency-only COA isn't enough for converted material.Was the lot screened for residual solvents and reaction-related impurities?
If the answer is vague, pause the purchase.Was remediation needed after conversion?
That doesn't automatically disqualify a lot, but it tells you the process likely needed additional correction.How does the supplier describe the aroma of the neat oil?
A serious manufacturer should be able to discuss organoleptic characteristics without hand-waving.
What works in real formulation
Converted cannabinoids can perform well in cartridges and concentrates when the cleanup is disciplined and the analytics are complete. In those cases, the distillate behaves like a stable base and lets your terpene design do its job.
What fails is buying converted oil as if it were a commodity. It isn't. The route to that oil affects flavor, stability, and your margin for error in formulation.
Sourcing and Supplier Due Diligence
A weak sourcing process creates more product problems than almost any terpene decision. If you're buying hemp-derived cannabinoids, supplier due diligence isn't administrative overhead. It's part of formulation.
The category grew quickly. As noted in Dad Grass's overview of hemp-derived THC, the U.S. Delta-8 THC market reached $2 billion by 2022, driven by conversion of surplus hemp CBD. That same growth is why serious buyers now demand more than a simple potency report before approving a cannabinoid input.

Read the COA like a manufacturing document
A COA should help you decide whether the material is safe, usable, and consistent. It shouldn't function as a sales brochure.
Start with these checks:
- Match the lot exactly: The lot number on the COA, sample label, invoice, and drum or jar should match. If they don't, stop there.
- Check the panel scope: For converted cannabinoids, potency alone isn't enough. You want a broader analytical picture.
- Review the date: Old data isn't useless, but it may not reflect the material you're receiving now.
- Look for method clarity: If the report is thin on testing detail, ask follow-up questions before release.
Red flags that deserve pushback
Some issues don't prove a batch is bad, but they do prove the review isn't finished.
| Red flag | What it suggests | What to do |
|---|---|---|
| Potency panel only | Incomplete screening for a converted input | Request a fuller panel before approving |
| Missing residual solvent data | The supplier may be avoiding the most relevant risk area | Hold the lot |
| Clean numbers with weak documentation | Reporting may be incomplete or recycled | Ask for traceable supporting records |
| No discussion of process route | Limited transparency on conversion history | Treat as higher risk |
| Sensory mismatch between lots | Variable cleanup or storage conditions | Requalify before blending |
A premium terpene blend can't rescue a cannabinoid input that should've been rejected in receiving.
Build a supplier file, not just a purchase trail
Strong teams don't evaluate each shipment in isolation. They build a record of how a supplier performs over time.
That file should include:
- COAs across multiple lots
- Any nonconformance notes
- Retention sample observations
- Communication quality
- Whether corrective actions were prompt and credible
If a supplier gives you complete data before you ask, answers directly, and stays consistent from lot to lot, that's useful. If every order requires chasing paperwork and decoding evasive answers, the low price usually stops being low.
A practical buying habit is to rank suppliers on two separate scales: analytical confidence and formulation suitability. Some vendors pass the first and fail the second.
If your team is still comparing vendors, this resource on where to buy terpenes from reliable suppliers is useful because the same sourcing discipline applies across aroma and cannabinoid inputs.
The standard that protects the brand
The right standard is simple. If a supplier can't give you enough information to defend the ingredient internally, don't put it into a product that carries your name.
That standard protects flavor consistency. It protects hardware performance. It also protects your customer support team from chasing complaints that started upstream in raw material purchasing.
Formulation Strategies for Hemp-Derived Inputs
Once the cannabinoid input is legally documented, analytically credible, and sensorially acceptable, the actual formulation work starts. Quality purchasing decisions prove their value at this stage.
The biggest practical issue is that converted cannabinoids don't just contribute potency. They also set the background your terpene system has to sit on. If that background is neutral, formulation is straightforward. If it carries residual process character, your top notes can flatten, your mid notes can get muddy, and your base notes may dominate in the wrong way.
Build the terpene structure around the base oil
A clean formula usually has a defined note structure:
- Top notes carry first impression. Citrus, pine, bright fruit, and other volatile components live here.
- Mid notes create identity. Floral, herbal, spice, and soft sweet notes often shape this zone.
- Base notes provide depth and persistence. Earthy, resinous, woody, and dense elements help the profile hold.
When the base cannabinoid oil is especially neutral, you can build with more precision. When it isn't, you need to compensate carefully rather than increasing total terpene load.
The Hemp Doctor's discussion of hemp-derived THC sourcing points to a critical blind spot for brands buying converted cannabinoids: residual conversion solvents may interact with or degrade terpene profiles. That's why supplier transparency and detailed lab analytics are so important before you ever start dialing in flavor.
Practical formulation habits that hold up
A few habits consistently improve outcomes:
- Run a neat-oil sensory check first: Smell and observe the cannabinoid input before adding anything. Don't wait until the final blend to discover a defect.
- Pilot small blends: Test your profile on the specific lot you're using. A terpene ratio that worked last month may not sit the same on a new distillate lot.
- Watch note balance, not just intensity: If top notes disappear too quickly, the issue may be the base oil or hardware pairing, not a lack of terpene concentration.
- Prioritize homogeneity: Uneven mixing creates inconsistent flavor and inconsistent customer experience.
- Keep records that tie sensory results to lot numbers: This gives you a formulation history you can effectively use.
The best terpene blend for distillate is the one that still tastes correct after it meets your real cannabinoid input, not the one that smelled best in the bottle.
For teams developing edible-adjacent, inhalable, or other ingredient systems that require cleaner flavor architecture, this guide on food grade terpenes for formulation work is a useful reference point for thinking about ingredient quality and application fit.
The broader answer to what does hemp derived mean is this. It means your cannabinoid source sits inside a specific legal framework, often follows a specific manufacturing route, and requires a more disciplined sourcing and formulation process than the label alone suggests. When you respect that reality, hemp-derived inputs can support stable, compliant, high-quality products.
If you're building cartridges, concentrates, or strain-inspired terpene systems and need reliable inputs for real production work, Gold Coast Terpenes offers lab-verified terpene blends, isolated compounds, and formulation resources designed for distillate optimization, flavor replication, and consistent batch performance.
