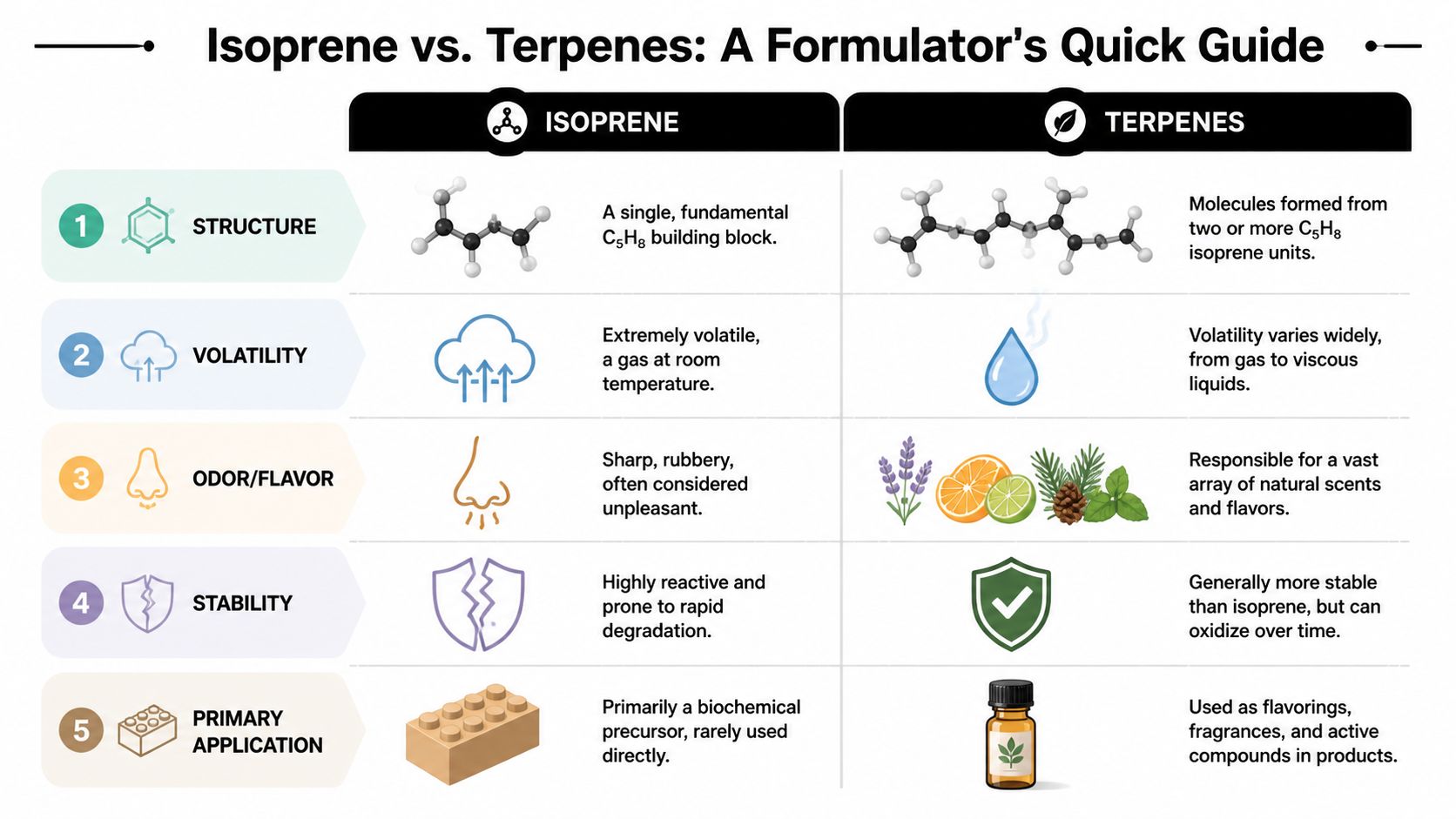
You’re reviewing a fresh CoA for a distillate batch meant for a strain-inspired terpene blend for vape cartridges. The expected peaks are there. Myrcene. Limonene. Maybe a little β-caryophyllene. Then there’s one extra signal you didn’t want to see: isoprene.
That small peak matters more than most junior formulators think. In commercial formulation, terpene vs isoprene isn’t a naming detail. It affects flavor accuracy, thermal behavior, blend stability, and whether your final cart tastes clean or develops a sharp, unwanted edge after filling and storage.
A lot of chemistry content stops at the textbook line that terpenes are built from isoprene units. That’s true, but it’s not enough to help you approve raw materials, troubleshoot an off-note, or decide whether a batch is fit for production. On the bench, free isoprene and finished terpenes play very different roles.
Why Isoprene vs Terpene Matters for Your Formulation
When you’re formulating for distillate, the target isn’t “more volatiles.” The target is the right volatile compounds in the right balance. Terpenes create the profile you can use. Free isoprene usually doesn’t.

A junior formulator will often see the word “isoprene” and assume it signals botanical authenticity because terpenes come from isoprene building blocks. That’s the wrong takeaway. In a finished vape formula, you’re not trying to enrich for precursor fragments. You’re trying to preserve a controlled sensory system made of monoterpenes, sesquiterpenes, and related compounds that behave predictably in oil.
What matters on the production side is simple:
- Flavor fidelity: A clean terpene blend preserves the intended top, mid, and base note structure.
- Thermal performance: The compounds need to behave well in cartridge operating temperatures.
- Batch consistency: Raw materials have to match the profile you spec, not just pass a broad volatile screen.
- QC clarity: Unexpected isoprene creates questions that slow release decisions.
Bench reality: If a profile smells flatter, harsher, or oddly rubbery after blending, check the chromatogram before you blame the hardware.
The Isoprene Rule and Terpene Biosynthesis
A common bench mistake starts with the right vocabulary and the wrong conclusion. A junior formulator sees “isoprene-derived” on a technical sheet and assumes any detectable isoprene-related signal supports authenticity. In practice, the opposite can be true. Free isoprene is the five-carbon precursor unit. The materials you build with are the terpene structures made from that unit and then modified into finished aroma compounds.

The isoprene rule gives you the structural logic. Terpenes are assembled from C5 units, so their carbon counts usually track in multiples of five. That framework is useful because it explains why monoterpenes, sesquiterpenes, and larger terpene classes behave differently in formulation, even before you get into oxidation, cyclization, or matrix effects in distillate.
How the building blocks become usable aroma compounds
Plants do not stop at making a free C5 fragment. They convert activated isoprene equivalents into longer chains, then reshape those chains into compounds with distinct odor, volatility, and stability profiles. That biosynthetic step is what turns a precursor concept into something commercially useful.
For formulation, three class-level distinctions matter:
- Monoterpenes are built from two isoprene units, giving a C10H16 backbone. Myrcene and limonene sit in this group.
- Sesquiterpenes are built from three isoprene units, giving a C15H24 backbone. β-caryophyllene is the familiar workhorse example.
- Higher terpene classes continue from the same rule, but they matter less in most vape blends unless you are working with heavier fractions or specialty aroma systems.
Those classes separate on more than naming. They differ in vapor pressure, odor impact, and thermal persistence. In practical terms, many monoterpenes leave the blend earlier, while many sesquiterpenes stay present longer and hold the profile together deeper into the puff sequence. You can verify compound-specific boiling behavior in reference databases such as PubChem entries for common terpenes like limonene and beta-caryophyllene, which is a better source for physical property checks than informal video summaries.
What that means during formulating
Inexperienced formulators often create avoidable problems. They learn that terpenes come from isoprene units, then treat any low-molecular-weight isoprenoid signal as part of the intended aromatic system. It is not. Biosynthetic origin does not make free isoprene a desirable formulation input.
A strain-style blend needs an evaporation curve you can control. Lighter monoterpenes drive the opening. Heavier sesquiterpenes support body and persistence. If the light fraction is too high, the cart peaks early and tastes hollow by mid-session. If the heavy fraction dominates, the profile can feel closed, dull, or resinous.
That same logic matters for contamination review. An unexpected isoprene peak is not just a chemistry footnote. It can point to raw material degradation, poor stripping during processing, or contamination introduced upstream. On the bench, that matters because it can distort sensory balance and complicate release decisions before the product ever reaches filling.
If you need a refresher on the plant-side pathway before these materials get isolated and blended, this overview of terpene biosynthesis in plants gives the background without losing the chemistry.
A good terpene profile is a controlled sequence of release and persistence. Free isoprene does not improve that sequence.
A Direct Comparison for Product Formulators
A junior formulator will often see “isoprene-derived” and assume anything in that family belongs in the blend. On the bench, that mistake shows up fast. The batch smells sharp, the front end flashes off too early, and the chromatogram raises questions you did not expect to answer during release.
| Property | Isoprene (C5H8) | Terpenes (C10+) | Significance for Formulation |
|---|---|---|---|
| Core structure | Single building block | Built from two or more isoprene units | Terpenes are the usable aroma ingredients. Isoprene is the precursor unit |
| Physical state tendency | Highly volatile small molecule | Broad range from light monoterpenes to heavier sesquiterpenes | Terpenes give you something you can design with across a volatility curve |
| Heat behavior | Boils at a very low temperature | Varies by class and structure | Heating changes release, flavor persistence, and hardware behavior differently |
| Sensory value | Unwanted in a finished blend | Drives targeted aroma and taste | Product quality depends on terpene identity and ratio, not generic volatility |
| Role in production | Possible impurity or degradation signal | Intended formulation component | Free isoprene belongs in a QC investigation, not in the target profile |
The practical difference is simple. Terpenes can contribute aroma, influence viscosity, and stay in the system long enough to matter during mixing and use. Free isoprene does not give you those benefits. Its boiling point is about 34°C, so it behaves more like a loss pathway or contamination flag than a design tool, as documented in the PubChem record for isoprene.
That distinction matters in commercial work because formulation is not just chemistry. It is fill consistency, sensory repeatability, and whether the product still presents correctly after time in hardware. A terpene blend lets you tune top, middle, and base behavior. An isoprene peak usually means you need to ask where it came from, whether from thermal stress, incomplete stripping, or upstream handling.
Terpenes also differ from each other in ways that matter more than new formulators expect. Monoterpenes can reduce viscosity and open the profile, but too much can make the formula feel thin or burn off early. Sesquiterpenes add body and staying power, but heavy use can mute brightness and slow wicking in some hardware. That is the trade-off you manage. For a closer look at why those classes behave differently, keep this chemistry of terpenes reference in your bench library.
Practical takeaways for product development:
- Use terpenes as functional blend components, not only flavor notes.
- Review any free isoprene signal as a possible impurity, degradation marker, or process residue.
- Balance volatility tiers instead of chasing only the loudest compounds in the bottle.
- Confirm performance two ways. Check the instrument result, then verify that the sensory profile holds through heating and repeated pulls.
Good formulators treat terpene selection as profile architecture. They treat isoprene as an exception that needs an explanation.
Why This Matters for Vape and Distillate Formulation
A formula can smell great in the bottle, pass a quick bench review, and still fail once it sits in a cartridge for two weeks. That is where new formulators get burned. Vape performance depends on how the terpene system behaves under heat, in contact with hardware, and over the product’s shelf life.

Terpenes do more than add flavor
In cartridge work, terpenes are formulation tools. They shape aroma, influence perceived strain identity, and affect how the oil presents during the first pull, the fifth pull, and the last third of the cart. That is why a proper terpene profile for distillate deserves the same discipline as cannabinoid potency or viscosity adjustment.
Build the profile with function in mind:
- Top notes create the first sensory hit and set brand expectation.
- Mid notes keep the profile intact through the body of the inhale.
- Base notes add persistence and keep the vapor from feeling hollow or washed out.
This is also why strain mimic work often misses the mark. Copying the loudest compounds from a COA does not recreate the experience in hardware. The result has to survive heating, repeated pulls, and storage without collapsing into a flatter, harsher profile.
What usually goes wrong in cartridge development
The recurring failures are predictable.
- Too much bright monoterpene content: The blend smells impressive at fill, then vapes sharp, thin, or short.
- Weak storage discipline: Oxygen exposure, warm hold conditions, and poor packaging control can shift the profile before the customer opens the box.
- Using total volatiles as a shortcut: A high aroma load does not tell you whether the blend is balanced or stable.
- Missing minor anomalies in review: Small unexpected peaks can explain off-notes, throat irritation, or drift far better than blaming the hardware.
Production rule: If a formula only performs in a fresh beaker and not in a filled cart, it is not ready for release.
Heat tolerance is part of the decision. Formulators use terpenes because they are the intended aroma compounds in the blend. Free isoprene is different. In a finished vape formula, it is not there to help profile construction, and it gives you another volatile variable that can show up as instability, off-character, or a process warning you failed to catch earlier.
That distinction matters even more in distillate, where the terpene fraction is doing a lot of sensory work with very little room for error. If you are building a profile for a specific oil type, this guide to the best terpenes for distillate is a useful starting point for matching blend intent to cartridge performance.
Isoprene as a Contaminant Risk in Terpene Blends
This is the point most chemistry explainers skip. They teach the isoprene rule, then leave the reader with the impression that any mention of isoprene is implicitly positive. For a finished vape formula, that’s backwards.
Free isoprene in a terpene blend is usually a contamination or degradation question, not a quality signal.
Where isoprene shows up in real processing
On the bench, unwanted isoprene can appear as a byproduct of extraction, terpene isolation, or thermal stress during processing. If a material sees too much heat, poor handling, or rough purification, small volatile fragments can show up where you wanted a cleaner terpene spectrum.
That matters most in profiles where nuance is doing the heavy lifting. In a fruit-forward blend, a trace off-note can flatten sweetness. In a gassy profile, it can push the formula from deliberate sharpness into a synthetic edge.
The practical problem is that teams often notice it late. They approve the batch because the major terpene peaks look right. Then the filled hardware comes back with complaints like harsh inhale, odd top note, or profile drift after a short hold.
What to do when you see an unexpected peak
Don’t overreact, but don’t wave it through either. Start with a disciplined check:
- Confirm it is isoprene and not a misread in the report.
- Compare with retained sample aroma instead of relying on the spreadsheet alone.
- Review process history for heat exposure, stripping conditions, or handling issues.
- Re-test if the profile is commercially important and you’re trying to preserve strain fidelity.
A useful practical reference here is that formulators frequently ask about “isoprene spikes” causing off-flavors, and that verifying purity with GC-MS to ensure isoprene levels are below 0.1% is critical for preserving aroma fidelity in complex profiles and meeting safety standards for vape products as summarized in this publication entry.
Free isoprene belongs in your risk review, not in your marketing language.
For junior formulators, the main lesson is simple. Don’t confuse “made from isoprene units” with “should contain measurable free isoprene.” Those are not the same thing.
Interpreting Lab Reports and Ensuring Purity
A batch lands on your bench with a CoA that looks clean at first glance. The headline terpenes are present, total terpene content is in range, and the supplier says it matches spec. That is still not enough to release it.
Read the report like a control document. A terpene blend succeeds or fails on identity, ratio, and unwanted trace components. Free isoprene matters here because it can hide behind an otherwise acceptable profile and show up later as harshness, top-note distortion, or poor stability in filled hardware.
What to read first on a CoA
Start with compound identity. Confirm that the dominant peaks fit the profile you are trying to build, then check whether the relative ratios make sensory sense for the intended application.
After that, review the minor peaks. Junior formulators frequently miss contamination, thermal breakdown, carryover from previous runs, or supplier cleanup problems when examining these peaks. A small unexpected peak may not move the total terpene number much, but it can still change how the blend performs in a vape or distillate system.
Use a fixed review sequence:
- Confirm the target terpene set: The expected compounds should be present in the right neighborhood, not just somewhere on the sheet.
- Check ratio logic: A believable profile has internal structure. Citrus-heavy, floral, gassy, and dessert profiles each tolerate different balance shifts.
- Flag unexplained peaks: Any unknown or out-of-place volatile needs a reason, especially if it suggests byproduct formation or poor fraction control.
- Compare against retained sensory samples: Instrument agreement with aroma is part of release, not a separate exercise.
Questions that prevent bad release decisions
| Review question | Why it matters in production |
|---|---|
| Do the major peaks match the target profile? | Confirms the batch is directionally correct before you spend time on fine detail |
| Do the minor peaks make chemical and process sense? | Catches contamination, degradation, or supplier process drift |
| Does the sensory result agree with the chromatogram? | Prevents approving a batch that looks acceptable on paper but fails in use |
| Is there any sign of free isoprene or another low-level volatile that should not be there? | Helps you stop off-note, harshness, and purity problems before filling |
One practical rule. Do not let total terpene percentage distract you from composition quality.
For incoming QC, I treat unexplained low-level volatiles as a supplier conversation, not a rounding error. If a peak is identified as isoprene, or even suspected to be isoprene, verify the method, review the lot history, and decide whether the blend is still acceptable for the product type you are making. A loose standard here creates expensive downstream failures.
If your team needs a tighter incoming inspection process, this guide to why purity matters in terpene quality standards is a useful reference for supplier qualification and batch release.
Formulator FAQs Terpene vs Isoprene
A lot of junior formulators ask the same questions once they start comparing lab data with sensory outcomes. These are the answers that matter in production.
Frequently Asked Questions
| Question | Answer |
|---|---|
| Is isoprene a terpene? | Not in the way formulators usually mean it. It’s the C5H8 building block used to form terpene structures, but it isn’t the same thing as the finished terpene compounds you use for aroma design. |
| If terpenes come from isoprene units, why is free isoprene a problem? | Because precursor logic and finished-product quality are different issues. A clean terpene blend should deliver specific intended compounds. Free isoprene can indicate byproduct formation, contamination, or degradation. |
| Which matters more for a vape formula, total terpene content or terpene identity? | Identity. A believable profile depends on which terpenes are present and how they’re balanced across volatility tiers. Total aroma load alone won’t give you strain accuracy. |
| How do I think about top, mid, and base notes in a strain-inspired terpene blend? | Lighter monoterpenes usually carry the opening. Mid-note compounds connect the profile. Heavier fractions, often including sesquiterpenes, keep the flavor from disappearing after the first pull. |
| Should I reject every batch that shows any isoprene? | Not automatically. First confirm the peak, compare with sensory results, and review process history. But you should treat it as a QC question that needs explanation, not as a premium feature. |
| What’s the right test method when I’m worried about contamination? | Use GC-MS when you need to verify purity and investigate suspicious peaks. It’s the practical tool for confirming whether an unexpected signal is affecting profile integrity. |
| Can isoprene help me replicate the flavor of a strain? | No. Replicating flavor of a strain depends on the right terpene composition and ratio, not on adding or tolerating free isoprene. |
| Why do some blends smell right in the bottle but wrong in the cart? | Usually because the volatility balance is off, the blend was overbuilt around loud top notes, or the material changed under heat or storage. Cartridge performance exposes weak profile architecture fast. |
The short version for junior staff
If you remember only four rules, keep these:
- Terpenes are formulation assets. Isoprene is not.
- A good chromatogram still needs sensory confirmation.
- Unexpected peaks deserve a process review.
- Profile structure beats raw intensity.
“Does this batch support the intended profile in actual hardware?” is the right question. “Does it contain a lot of volatiles?” isn’t.
If you’re sourcing inputs for formulating strain-inspired terpene blends for vape cartridges or distillate, Gold Coast Terpenes offers strain-specific profiles, isolated compounds, and formulation resources that can help you build cleaner, more consistent products. Their catalog is useful for teams that need THC-free terpene options, batch-tested materials, and practical tools for dialing in flavor accuracy without relying on filler diluents.
