If you've ever heard that THC boils at 157°C (315°F), you've been told a half-truth that can cause major headaches for product formulators. That number is technically correct, but only in a near-perfect vacuum—a condition you'll never encounter during extraction, distillation, or inside a vape cartridge.
Getting this right is a foundational step to mastering your process, ensuring product consistency, and achieving target flavor profiles.
What Is the Real Boiling Point of THC?
The honest answer is: there isn't just one. The boiling point of THC isn't a fixed temperature; it’s a variable that shifts based on the ambient pressure. Understanding this relationship is critical for any formulator or extractor.
The Pressure-Temperature Relationship
Think about boiling water. It boils at 100°C at sea level, but on a mountain where atmospheric pressure is lower, its boiling point decreases. Cannabinoids like THC behave in the exact same way.
When you lower the pressure in your equipment, you make it much easier for THC molecules to escape the liquid phase and enter the vapor phase. This is the core principle behind modern cannabis distillation. You pull a vacuum to lower the boiling point, allowing you to purify the oil without thermal degradation.
This chart illustrates how dramatic the difference is.
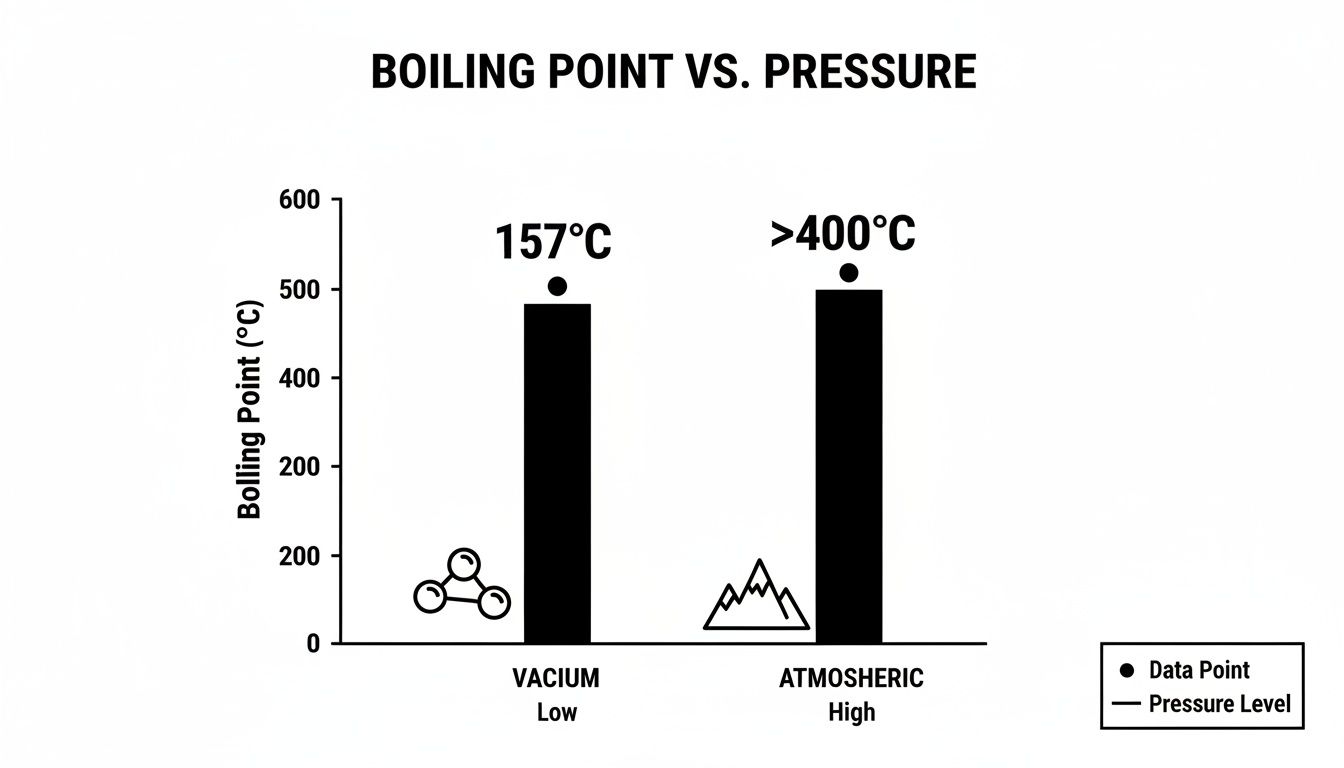
As you can see, attempting to boil THC at normal atmospheric pressure is a recipe for disaster. It's why running a distillation setup without a proper vacuum is a non-starter for producing high-quality distillate.
Practical Temperature Ranges for Formulation
At standard atmospheric pressure (760 Torr), you would need to reach over 400°C (752°F) to get THC to boil. At that temperature, you're not just boiling the THC—you're completely destroying it, along with every valuable terpene and cannabinoid.
However, when you pull a deep vacuum with a short-path distillation system, the game changes. Under a vacuum of around 0.02 Torr, the boiling point drops to a much more manageable 200°C (392°F). This is the temperature range where purification happens.
Key Takeaway: The goal isn’t to chase a single magic number. It’s to use your vacuum pump to bring the boiling point into a safe and effective window—usually between 160°C and 200°C—where you can separate and purify THC without degrading it.
The table below breaks down exactly how pressure affects the boiling point of Delta-9-THC, making it clear why vacuum is essential for any serious extraction operation.
THC Boiling Point at Different Pressures
| Pressure Level | Pressure (mm Hg / Torr) | Approximate Boiling Point (°C) | Approximate Boiling Point (°F) |
|---|---|---|---|
| Standard Atmosphere | 760 | >400°C | >752°F |
| Rough Vacuum | 1.0 | 243°C | 469°F |
| Medium Vacuum | 0.1 | 210°C | 410°F |
| High Vacuum (Short Path) | 0.02 | 200°C | 392°F |
| Ultra-High Vacuum | 0.001 | 157°C | 315°F |
This relationship is everything when it comes to making high-quality distillate.
By carefully controlling your vacuum level, you gain precise control over temperature. This allows you to selectively boil off and separate THC from heavier compounds like waxes, lipids, and chlorophyll. Understanding this process is fundamental to creating the clean, potent base oil needed for premium vape and edible formulations.
To learn more about how this applies to production, check out our guide on how to make THC oil.
How Pressure Changes the Game for THC Vaporization

If you've spent any time trying to purify cannabis oil, you know that the relationship between pressure and temperature is everything. It’s the one concept that, once mastered, separates professional-grade production from amateur efforts.
Think of it like this: trying to vaporize THC at normal atmospheric pressure is like trying to push your way out of a packed concert. There’s a dense "crowd" of air molecules constantly pressing down on your oil. To get the THC molecules to "escape" as a vapor, you have to apply an enormous amount of energy—in this case, scorching hot temperatures that will degrade your final product.
That atmospheric resistance is your enemy. It forces you to use destructive levels of heat. This is precisely why working under a vacuum isn't just a good idea; it's essential for high-quality extraction.
Putting the Vacuum to Work
When you connect a vacuum pump to your distillation apparatus, you are literally removing that "crowd" of air molecules from the system. With far less pressure holding them down, THC molecules can break free from the liquid and become a vapor with much less thermal energy.
This is the entire principle behind modern techniques like short-path and wiped-film distillation, which are the gold standard for creating pure, clean distillate. You’re not just boiling the oil. You're creating a controlled environment where THC can be gently encouraged to vaporize at temperatures low enough to prevent it from burning or degrading.
A Quick Analogy: Think of the air pressure in your lab as a heavy lid sitting on top of your oil. A vacuum pump lifts that lid, letting the cannabinoids vaporize easily without you having to crank the heat up to damaging levels.
This is how you transform raw, crude oil into the beautiful, transparent distillate that top-shelf products are made of. Mastering the vacuum gives you an incredible amount of control over the entire separation process.
Finding the Sweet Spot with Pressure Control
For anyone formulating cannabis products, this is where you go from just following steps to truly engineering your extracts for quality and consistency. The real goal is to find that perfect balance—a vacuum deep enough to drop THC's boiling point into a safe and effective range, which is typically between 160°C and 200°C.
This temperature window is the sweet spot. It's low enough to prevent thermal degradation but hot enough to efficiently separate THC from all the undesirable components you don't want in your final product.
Things you leave behind include:
- Waxes and Lipids: These heavy compounds have sky-high boiling points, so they stay behind as residue.
- Chlorophyll: This plant material is easily separated out during distillation.
- Degraded Cannabinoids: By keeping temperatures low, you prevent the formation of compounds like CBN that can result from overheating.
By carefully dialing in your vacuum, you can selectively pull the THC vapor away from these impurities, leaving you with a much cleaner and more potent oil. This precision is also crucial for preserving the fragile terpenes that give your product its signature aroma and flavor.
If you want to get a better feel for the machinery involved, our guide on how to make cannabis concentrates is a great place to start. Just remember, pressure isn’t simply another setting to tweak. It's the primary tool you have for controlling the purity, potency, and overall quality of your extract.
Temperature Control for Decarboxylation and Distillation
Getting temperature right isn't about finding one magic number. It's about mastering a sequence of carefully controlled heat applications throughout your entire production process. As a formulator, you'll find that THC's boiling point is most critical during two key stages: decarboxylation and distillation. Each one demands a unique heating strategy to achieve the target potency and purity.
Nailing the right heat at the right time is what separates a premium, water-clear distillate from a dark, degraded, or weakly activated final product.

Optimizing Decarboxylation Temperature and Time
Before you can distill THC, you have to activate its raw, acidic form, THCA. This is what decarboxylation is all about—gently heating your crude oil to convert the inactive THCA into active THC by removing a carboxyl group. It’s a delicate dance between time and temperature.
The goal here is simple: achieve full conversion without overshooting your mark and degrading that precious THC into CBN. Too much heat for too long yields a product with unwanted characteristics and a compromised flavor profile. While there's no single "perfect" setting, a reliable range for decarbing is 110–120°C (230–250°F).
Holding your oil in this sweet spot for 30 to 60 minutes typically achieves nearly full conversion. You can monitor the reaction visually; the oil will bubble as it releases CO2, and when that bubbling slows to a stop, you know the process is near completion. Rushing the process by increasing the heat is a classic mistake that leads to significant potency loss. If you want to dive deeper, our guide on how to properly decarb oil for vape cartridges provides more detail.
Navigating Distillation with Vacuum and Heat
Once your oil is fully decarbed, it's time to purify it through distillation. This is where the relationship between THC's boiling point and pressure truly comes into play. As we’ve covered, trying to boil THC at normal atmospheric pressure would completely destroy it. That's why we use equipment like wiped-film and short-path systems, which create a deep vacuum to lower the boiling point into a safe, workable range.
In a distillation run, you're essentially separating the THC from everything else. The process unfolds in a few key stages:
- Stripping Volatiles: First, you’ll do a low-temperature pass (around 130–150°C under vacuum) to pull off any leftover solvents and the most volatile terpenes.
- The THC Fraction: Now for the main event. You’ll raise the temperature into the 160–200°C range. This is where THC vaporizes and collects on a condensing surface, leaving the heavier, undesirable compounds behind.
- Removing Heavies: Waxes, lipids, and chlorophyll have much higher boiling points, so they never vaporize and are left behind as residue.
The debate around THC's true boiling point really heated up around 2010 with the boom in personal vaporizers, leading to a lot of conflicting numbers. For example, some sources claim 315°F (157°C) is the target, but that’s only possible under a near-perfect vacuum. For extractors in the U.S.—who handle an estimated 80% of global distillate production—practical work in short-path systems at 0.1 mm Hg keeps temperatures under 160°C, preserving over 90% potency. The risks are real: improper decarbing below 200°F might only activate 50% of the THC, while overheating above 410°F can degrade 15–20% of it into CBN.
By precisely controlling both the vacuum depth and the evaporator temperature, formulators can cleanly "fraction off" the THC. The result is a highly potent, crystal-clear, and flavorless distillate—the perfect blank canvas for reintroducing specific terpene profiles to create consistent, high-value products.
Formulating Flavor with Terpene Boiling Points
Crafting a superior vape cartridge is about engineering the user experience. It's not just about delivering THC into the vapor; it’s about controlling what vaporizes and when. This is where the art and science of boiling points come into play, allowing you to build a flavor journey from the first puff to the last.
The key is understanding the different boiling points of your cannabinoids and the terpenes you're blending into your distillate. By using temperature as a tool, you can create a specific sequence of aromas and flavors that makes your product stand out.
Building a Sequential Flavor Release
While cannabinoids and terpenes form a complex vapor together, the basic rules of boiling points still apply. The most volatile compounds—those with the lowest boiling points—will always vaporize first, defining the initial impression.
You can think of it like building a fragrance:
- Top Notes (The First Impression): Highly volatile terpenes like Myrcene and Limonene have low boiling points. They create that initial burst of flavor and aroma that the user tastes immediately. This is your hook.
- Mid Notes (The Body): Next up are terpenes with a medium boiling point, like Linalool and Humulene. They build on the top notes, adding complexity and creating a more rounded, full-bodied flavor profile.
- Base Notes (The Foundation): Finally, the cannabinoids like THC and the heavier, less volatile terpenes vaporize. These deliver the core effects and the deep, lasting flavor notes that tie the whole experience together.
This layered approach gives you incredible control. You can design a vape that kicks off with an energetic citrus blast from Limonene before settling into the calming, earthy effects of the THC and other terpenes.
From Raw Ingredients to a Finished Experience
To execute this effectively, you must know your ingredients. Terpenes like Beta-Caryophyllene (130°C), Myrcene (167°C), and Limonene (176°C) have relatively low boiling points. When creating a blend for a vape device operating at a common temperature like 350°F, these are the compounds that will define the initial character of your product.
We now know for a fact that these lighter monoterpenes are inhaled before the cannabinoids in a vaporizer. This simple scientific principle is why 70% of users say they prefer a vape temperature between 360-390°F—it’s the sweet spot that perfectly balances rich terpene flavor with potent cannabinoid activation.
In markets like Europe and Australia, where terpene-rich products make up 40% of sales, getting this right is huge. In fact, precisely matching your terpene profile to the THC vaporization point has been shown to cut down on perceived harshness by as much as 25%.
Formulator's Tip: When you're sketching out a new blend, ask yourself what you want the first impression to be. A sweet, fruity hit? Or something more earthy and spicy? Select top-note terpenes with lower boiling points to create that first wave of flavor, and then build the rest of your profile from there.
The table below is a great starting point, giving you the boiling points for some of the most common compounds you'll be working with. For a deeper dive, check out our complete terpene temperature chart for formulators.
Boiling Points of Common Cannabinoids and Terpenes
Here’s a quick reference guide for the approximate boiling points of key compounds at standard atmospheric pressure. This is essential knowledge for designing vape and concentrate profiles that deliver on their promise.
| Compound | Type | Boiling Point (°C) | Boiling Point (°F) | Associated Aroma/Effect Note |
|---|---|---|---|---|
| Beta-Caryophyllene | Terpene | 130°C | 266°F | Spicy, peppery (Base Note) |
| Alpha-Pinene | Terpene | 156°C | 313°F | Pine, sharp (Top Note) |
| Myrcene | Terpene | 167°C | 333°F | Earthy, musky (Mid Note) |
| Limonene | Terpene | 176°C | 349°F | Citrus, sweet (Top Note) |
| Linalool | Terpene | 198°C | 388°F | Floral, lavender (Mid Note) |
| Delta-9-THC | Cannabinoid | ~200°C | ~392°F | Core psychoactive effect |
| CBN | Cannabinoid | 185°C | 365°F | Sedative effect note |
| CBD | Cannabinoid | 180°C | 356°F | Non-psychoactive effect |
| Terpineol | Terpene | 218°C | 424°F | Lilac, floral (Base Note) |
Once you start working with this data, you stop just mixing ingredients and start truly designing a product. You can protect those delicate, expensive top-note terpenes from getting burned off while making sure your cannabinoids are vaporized properly for a consistent, high-quality experience every single time.
How to Avoid Degradation and Unwanted Byproducts
Incorrect temperature control is an expensive mistake. While heat is necessary to purify THC, mishandling it at any stage can ruin an entire batch of oil. When you apply excessive heat, you’re not just destroying potency—you're creating unwanted byproducts that degrade the flavor, compromise safety, and damage your brand's reputation.

The name of the game is avoiding degradation. Pushing cannabinoids and terpenes past their thermal limits sets off a chain reaction of chemical changes, and none of them are desirable. Let’s look at what can go wrong and how to prevent it.
The Conversion of THC to CBN
One of the most common signs of poor processing is the conversion of valuable Delta-9-THC into Cannabinol (CBN). CBN is not naturally synthesized by the plant in significant quantities; it is a degradation product of THC. High levels of CBN in a final product are a red flag for poor process control or aged starting material.
CBN has a boiling point of 185°C (365°F), which is lower than THC's. When you apply excessive heat during decarboxylation or distillation, you accelerate the oxidation that converts your primary active cannabinoid into this unwanted byproduct.
This creates two major problems for your final product:
- Reduced Potency: Every molecule of THC that degrades into CBN is a direct loss of your target active ingredient.
- Altered Effects: CBN is known for its sedative properties. A high-CBN product will not deliver the intended user experience and often leads to feedback about feeling tired or groggy.
Burning Terpenes and Creating Off-Flavors
Even more sensitive to heat than cannabinoids are terpenes. These delicate compounds are responsible for the product's signature aroma and flavor. When you scorch them, you don't just lose those desirable notes—you create harsh, unpleasant ones in their place.
When a delicate terpene like Limonene or Myrcene is overheated, it can create a burnt, acrid taste that completely ruins the intended flavor profile. This is a primary reason for customer complaints about a vape cartridge tasting "harsh" or "chemical."
Protecting your terpenes is non-negotiable. It all comes down to choosing the right vape hardware with precision temperature settings and respecting the thermal limits of your specific terpene blend. Our guide to formulating with strain-inspired terpene blends dives deeper into creating formulations that are both stable and flavorful.
Actionable Tips for Process Control
Since the cannabis legalization waves in markets like California (2016) and Canada (2018), getting the THC boiling point right has shaped a global vape market now worth over $30 billion. Yet, the data is sobering: a staggering 65% of cartridge failures stem from overheating, which can burn off 10-25% of the cannabinoids. These are costly, avoidable mistakes. You can discover more insights about these industry trends on cbdsloth.com to get a better handle on the numbers.
To keep your product from becoming another statistic, here are a few best practices to implement:
- Calibrate Your Equipment: Make it a habit to regularly check the temperature probes and controllers on your decarb reactors and distillation gear. Don't trust, verify.
- Select Quality Hardware: Opt for vape cartridges that use ceramic heating elements and pair them with batteries offering variable voltage. This gives you (and your customers) far better control over the heat.
- Respect Thermal Limits: Always build your formulation around the boiling points of your most fragile terpenes. If you have a low-temp terpene blend, it needs a device that can deliver a low-temp experience.
Matching Vape Hardware to Your Formulation
You can spend weeks perfecting a formulation, but if you put it in the wrong hardware, all that work is wasted. Think of it this way: your oil is the engine, but the cartridge is the vehicle. A poorly matched device will burn your flavors, waste your product, and leave customers with a negative experience.
The goal is to create a single, cohesive system where the oil and the device work together flawlessly. This means you have to think about hardware from the very beginning, seeing it as a crucial part of the formulation process itself.
Choosing the Right Heating Element
At the heart of every vape is the heating element, often called an atomizer. This one component has the biggest impact on how evenly your oil heats up and whether you preserve those delicate terpenes you worked so hard to include.
You'll encounter three main types of coils on the market today:
- Wick Systems: These are the old-school standard, using silica wicks to pull oil onto a metal coil. They are notorious for creating hot spots that scorch the oil, leading to harsh, burnt flavors.
- Quartz Coils: Quartz heats very quickly. While that means rapid vapor production, it can also spike to temperatures that flash-vaporize the most volatile terpenes, eliminating the top notes of your flavor profile.
- Ceramic Coils: For good reason, ceramic has become the gold standard for premium products. Its porous structure absorbs the oil and heats it gently and evenly. This provides a lower, more consistent temperature that is ideal for allowing a complex terpene profile to be fully expressed.
For any formulation that is rich in terpenes, a ceramic coil is almost always the correct choice. It provides the gentle, controlled heat needed to vaporize the entire spectrum of your blend without destroying the flavor.
Understanding Resistance and Voltage
Beyond the material, you need to be familiar with the electrical characteristics of the hardware. The coil's resistance (measured in ohms, Ω) and the battery's power (measured in voltage, V) work together to determine the final temperature your oil experiences.
Key Concept: A low-resistance coil paired with a high-voltage battery gets hotter, faster. Conversely, a higher-resistance coil requires less voltage to reach the optimal vaporization temperature, giving you much finer control.
This is where your job as a formulator extends beyond the lab. If you've crafted an oil loaded with delicate, low-boiling-point terpenes, you must pair it with hardware that can handle it properly.
- For High-Terpene Formulations: Your best bet is to recommend higher-resistance coils (1.2-1.6Ω) paired with variable-voltage batteries set low (around 2.5-3.0V). This prevents scorching and lets the full flavor profile unfold gradually.
- For High-Viscosity Distillates: Thicker oils sometimes require more power to vaporize efficiently. A lower-resistance coil (0.8-1.2Ω) can work, but it’s vital to instruct customers to use short, controlled draws to avoid overheating the oil.
Ultimately, your responsibility doesn’t end when the oil is mixed. Providing clear guidance on which battery to use—and what settings to use—ensures consumers experience your product exactly as you intended. It protects their experience and, just as importantly, your brand's reputation. For a deeper look at matching oils and hardware, our terpene formulation guide is a great next step.
Frequently Asked Questions About THC Formulation
Theory is one thing, but when you're in the lab, practical questions always arise. Let's walk through some of the most common issues formulators face and provide clear, actionable answers.
What Is the Most Common Mistake When Heating Distillate?
The most common mistake is applying excessive heat when mixing terpenes back into a distillate. The rationale is that high heat is needed to reduce the oil's viscosity for a homogenous mix, but this approach is counterproductive.
It torches your most volatile—and expensive—top-note terpenes. You end up destroying the very flavor profile you were trying to create.
Gentle heat is your best friend here. Aim for a homogenization temperature around 50–60°C (122–140°F). It’s just enough warmth to loosen up the oil for a perfect, even blend without cooking off your precious terpenes.
Why Do My Vape Cartridges Taste Burnt?
A burnt taste is almost always a heat management problem. However, the culprit can be either your formulation or your hardware.
If a formulation is too viscous, the wicking material can't absorb the oil fast enough between draws, causing it to run dry and burn. The other common issue is mismatched hardware that runs too hot, scorching the oil on contact with the heating element.
This is exactly why knowing the boiling point of THC and your specific terpene blend is so critical. A formulation rich in low-boiling point terpenes needs a device that operates at lower temperatures, such as a quality ceramic coil paired with a variable-voltage battery. Our guide on replicating the flavor of specific strains dives deeper into creating stable blends that work with your hardware, not against it.
How Does Decarboxylation Affect the Boiling Point of THC?
This is an excellent question because it gets to the heart of process control. Decarboxylation itself does not change THC's boiling point. Instead, think of it as a crucial, temperature-sensitive preparatory step.
During "decarb," you are gently heating crude oil to convert the non-psychoactive THCA into active THC. If you overshoot the temperature—pushing past 130°C (266°F)—you will begin to degrade that newly formed THC into CBN and vaporize valuable terpenes present in your crude extract.
Proper decarboxylation ensures you have the maximum amount of THC available for the main event: distillation. That is the stage where controlling the boiling point under vacuum becomes your top priority. For a more detailed walkthrough, check out our terpene formulation guides.
At Gold Coast Terpenes, we provide the lab-verified, strain-specific terpene profiles and isolates you need to achieve consistent, high-quality results in every batch. Elevate your formulations with precision-blended components designed for professionals. Explore our full catalog at https://www.goldcoastterpenes.com.
