For formulators, extractors, and manufacturers, understanding terpene boiling points isn't academic—it's a commercial necessity. These values directly impact the flavor accuracy of a vape cartridge, the consistency of a distillate, and the stability of a final product. Mastering temperature control during formulation is essential for driving product quality, ensuring batch-to-batch consistency, and protecting your bottom line.
Why Terpene Boiling Points Are Key to Quality Formulation
In cannabis product development, precise temperature control is often the deciding factor between a premium, stable product and a failed, degraded batch. This isn't just about chemistry; it's about protecting your investment in high-quality isolates and blends. A solid grasp of when and how different terpenes vaporize is required to preserve the exact flavor and aroma profile you designed for your product.
The Commercial Impact of Temperature
Consider your terpene blend as a structured aromatic profile. The lighter, more volatile monoterpenes are your bright top notes. The heavier, less volatile sesquiterpenes are the deep, earthy base notes that provide depth and longevity. Applying excessive heat during mixing or formulation will volatilize and "burn off" these delicate top notes before they are successfully integrated into the final product, leaving you with a dull, one-dimensional flavor.
Understanding the unique boiling point of terpenes allows you to design a heating protocol that respects each compound. This is critical for several key formulation goals:
- Replicating Strain Profiles: To accurately replicate the profile of a strain like OG Kush, you must preserve the precise ratio of its constituent terpenes. If the myrcene is degraded due to improper heating, the final product will fail to meet the target profile.
- Formulating for Vape Cartridges: When mixing terpenes into a viscous distillate, sufficient heat is needed to achieve a homogenous blend. However, too much heat will destroy the very compounds responsible for the product's flavor and aroma.
- Ensuring Flavor Consistency: Customers expect a consistent experience with every purchase. Controlling for terpene boiling points is how you deliver that reliable flavor and aroma, batch after batch, strengthening brand loyalty.
While cannabinoid potency is a key metric, aroma and taste are primary drivers of consumer purchasing decisions. Failing to protect your terpene profile during formulation is a critical error that undermines product quality and market competitiveness.
This guide provides the practical data needed for precise cannabis product formulation. We will examine how boiling points influence your strategy and differentiate professional-grade products from the rest. While the science of terpenes is our focus, even factors like starting material genetics play a role. You can learn more about male vs female plant characteristics to see how deep the science goes. Mastering these fundamentals is essential for building a trusted brand in a competitive market.
The Science of Terpene Volatility and Vapor Pressure
To master terpene formulation, you must look beyond boiling points and understand volatility. A boiling point is the specific temperature at which a liquid becomes a vapor. Volatility, however, describes a substance's tendency to evaporate, which can occur at temperatures far below its boiling point.
This distinction is critical for product formulation. A common error is assuming terpenes are stable as long as temperatures remain below their boiling points. The reality is that significant aromatic loss begins long before that threshold is reached. This gradual evaporation is driven by a property called vapor pressure.
What Vapor Pressure Means for Your Bottom Line
Think of vapor pressure as the "escape energy" of a terpene molecule. It's the pressure exerted by vapor molecules in equilibrium with their liquid state. As temperature increases, so does this pressure. Even a minor increase in ambient lab temperature can accelerate the loss of valuable aromatic compounds.
Lighter monoterpenes, such as Pinene and Limonene, have higher vapor pressures than heavier sesquiterpenes like Beta-Caryophyllene. This property makes them the bright, impactful "top notes" of a flavor profile. It also means they are the first to evaporate from a blend during mixing or even from an improperly sealed container in storage. Losing these compounds results in a product that tastes flat and lacks the initial aromatic "pop" you engineered.
The most significant terpene loss often doesn't happen in a single high-heat event. It's a slow, silent degradation that occurs whenever terpenes are exposed to air. Managing volatility is about controlling a constant state of evaporation, not just avoiding boiling.
The Real Cost of Terpene Evaporation
This is not just theoretical; it has a direct financial impact. The degradation process begins at surprisingly low temperatures, with some of the most volatile compounds starting to evaporate at just 70°F (21°C). Studies have shown that improper handling at room temperature can lead to a 10-30% loss of monoterpene content—a costly depletion of the very ingredients that differentiate your product.
For formulators using our complex strain-inspired terpene blends, preserving every delicate note is essential for achieving a true-to-strain profile. You can explore the research on terpene degradation at lower temperatures to see just how sensitive these molecules are.
This relationship between vapor pressure and temperature is why a batch of distillate left open on a lab bench quickly loses its aromatic character. As ambient temperature rises, more terpenes convert to vapor and dissipate, altering your carefully designed profile.
When developing products—especially for vape cartridges—strict temperature control during storage, transport, and formulation is not just best practice. It is an essential component of protecting your investment and ensuring the final product meets commercial quality standards.
A Comprehensive Terpene Boiling Points Chart
For formulators, knowing the boiling point of each terpene is foundational to creating consistent, repeatable products. This data dictates your heating protocols, from homogenizing terpenes into distillate to ensuring the intended sensory experience in a finished vape cartridge. Without these numbers, you risk scorching expensive terpenes and delivering an inconsistent product.
To support your formulation efforts, we've compiled a practical chart of common terpenes, organized by their chemical class and their role as top, middle, or base notes. This framework helps you move beyond a simple list of temperatures and start constructing balanced, layered flavor profiles.
Monoterpenes: The Bright Top and Mid Notes
Monoterpenes are the lighter, more volatile compounds in your blend. They possess lower boiling points and higher vapor pressures, meaning they are the first aromas to be perceived. Preserving these is key to a product's initial impact.
These are your top notes—the vibrant, defining scents of a strain's character. Their low boiling points also make them highly susceptible to degradation from excessive heat. Careful temperature control is a requirement when working with these delicate molecules.
- α-Pinene: Boiling at 155°C (311°F), this terpene provides a sharp, fresh pine aroma. As a quintessential top note, it requires a gentle heating protocol to preserve its signature scent.
- Myrcene: With a boiling point of 167°C (333°F), Myrcene is a versatile mid-note. It is excellent for bridging the gap between bright top notes and earthy base notes.
- d-Limonene: Known for its bright citrus aroma, Limonene boils at 176°C (349°F). Proper handling of this mid-note is essential for fresh, uplifting profiles. For a reliable starting point, our pure d-Limonene isolate offers excellent consistency.
- Linalool: With a boiling point of 198°C (388°F), Linalool provides a floral mid-note. Its relative stability makes it more forgiving than more volatile monoterpenes.
This chart illustrates the wide range in boiling points, from the highly volatile Isoprene to the more robust Myrcene and Geraniol.
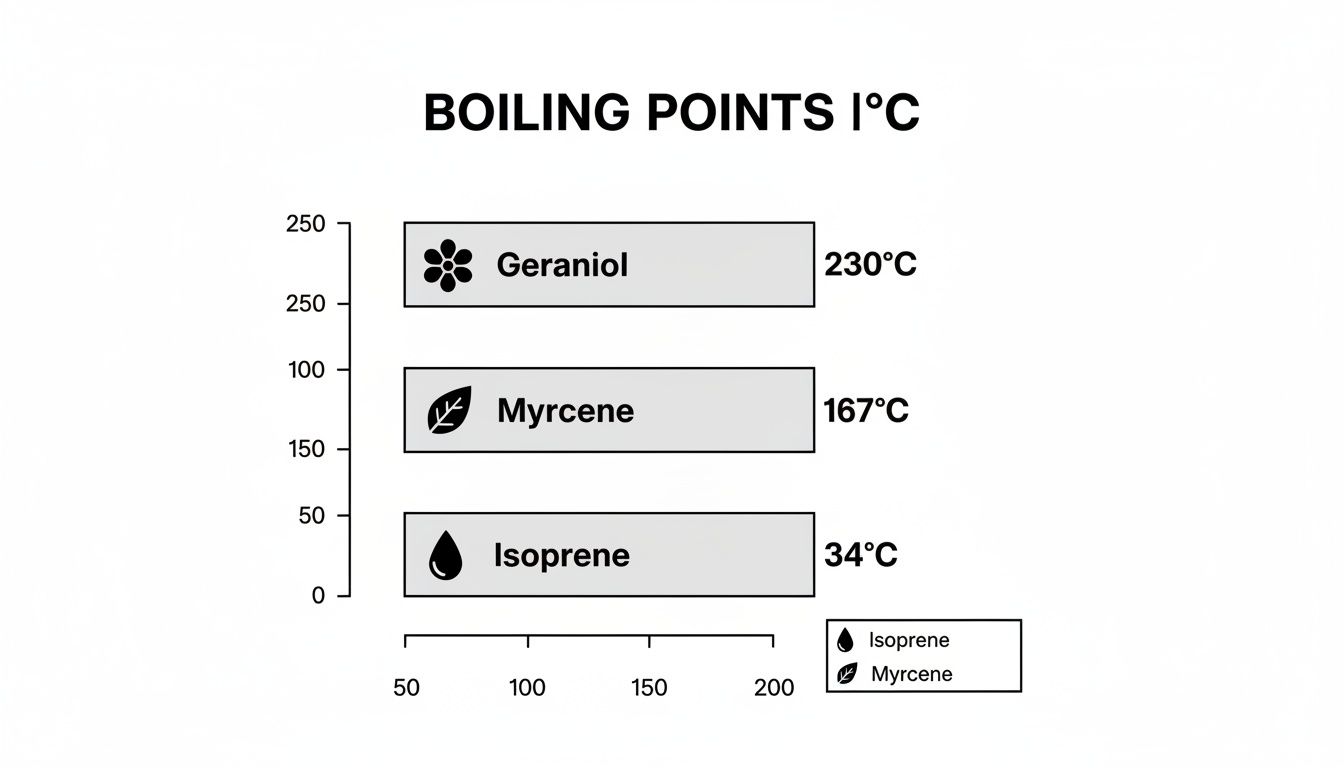
This is a clear visual reminder of why a "one-size-fits-all" heating approach is ineffective. The most delicate top notes will be lost long before heavier compounds have properly homogenized.
Sesquiterpenes: The Rich Mid and Base Notes
Sesquiterpenes are heavier, more complex molecules with significantly higher boiling points. In formulation, they function as base notes, providing the deep, lingering aromas that give a blend its full-bodied character and longevity. They also act as fixatives, helping to anchor the more volatile monoterpenes.
- β-Caryophyllene: Boiling at a verified 263°C (505°F), Caryophyllene contributes a spicy, peppery base note. Its high boiling point means it can withstand higher processing temperatures, making it a robust foundation for complex blends.
- Humulene: This terpene has an earthy, woody aroma and boils at 198°C (388°F). It is an excellent mid-to-base note that adds depth and complexity to a profile.
- Nerolidol: With a boiling point of 276°C (529°F), Nerolidol provides a subtle, woody-floral base. Its stability makes it an effective fixative for anchoring volatile top notes and extending the life of the overall flavor. Our formulation resources offer more guidance on building these kinds of sophisticated profiles.
Let's correct some persistent misinformation that impacts product formulation. In this industry, accurate data is your most valuable asset. Inaccurate data leads to ruined batches, wasted capital, and inconsistent products.
The Beta-Caryophyllene Myth
Many online terpene charts and older formulation guides list the boiling point for Beta-Caryophyllene around 119°C (246°F) or 130°C (266°F). This information is incorrect and is a classic example of how misinformation can become entrenched.
The scientifically verified boiling point of Beta-Caryophyllene is 263°C (505°F). This is a difference of over 130°C. This is not a rounding error; it's a massive discrepancy with significant implications for anyone replicating the flavor of a specific strain or perfecting a custom blend for distillate.
The error surrounding Beta-Caryophyllene’s boiling point is a critical lesson in data verification. Always vet your sources and rely on scientifically proven figures. It is the only way to build reliable SOPs and avoid costly formulation mistakes.
Why Accurate Data Matters
Knowing the true boiling point allows for more effective and efficient product manufacturing. The confusion has led many formulators to be overly cautious, using processing temperatures that are too low out of fear of degrading this key compound. This has likely resulted in poor homogenization, reduced throughput, and inconsistent final products.
When developing strain-inspired terpene blends, understanding that Beta-Caryophyllene can withstand higher temperatures is a game-changer. With the correct data, you can confidently process blends at more effective temperatures without risk of degradation. This adjustment can improve efficiency and yield. This situation underscores the importance of working with a supplier like Gold Coast Terpenes, a team that verifies every terpene profile for cannabis product formulation.
Putting Temperature Data to Work in Your Lab
Knowing a terpene's boiling point is interesting, but applying that knowledge to produce better, more consistent products is what matters commercially. Let's translate this data from the chart to the lab bench to optimize distillation and vape cartridge formulation.
Fine-Tuning Distillation and Separation
For extractors, temperature is the primary tool for fractional distillation and purification. Because each terpene has a unique boiling point, you can use temperature ramps to selectively capture or remove specific aromatic compounds.
For example, to retain light top notes like α-Pinene (BP: 155°C / 311°F), a slow, gradual increase in temperature allows you to "pull off" and collect these delicate terpenes first for reintroduction later. Conversely, when isolating a resilient sesquiterpene like Beta-Caryophyllene (BP: 263°C / 505°F), you can confidently increase the heat after the more volatile compounds have been safely collected.
Precise temperature control during distillation is about strategic preservation. By understanding the boiling point of terpenes, you can design a heating protocol that deconstructs and then reconstructs a profile with maximum fidelity.
Optimizing Vape Cartridge Formulation
The primary challenge in formulating for vape cartridges is homogenizing viscous cannabinoid distillate with a liquid terpene blend without scorching the aromatics. The goal is to find the "sweet spot" temperature: hot enough to lower the distillate's viscosity for mixing, but cool enough to remain well below the boiling point of your most delicate terpenes.
A common mistake is improper heating, resulting in a final product that smells nothing like the terpene blend that was introduced. Of course, even with a perfect profile, other factors can come into play. For a deeper look at how genetics influence a final product, you can read our guide on the Sour Diesel strain.
Here is a repeatable framework for your lab:
- Gently Warm the Distillate: Begin by heating your cannabinoid concentrate alone in a sealed container to a target temperature of 65–75°C (150–167°F). This lowers viscosity without approaching temperatures that would damage terpenes.
- Remove from Heat: This step is critical. Never add terpenes to a distillate that is actively being heated. Once the oil reaches its target temperature, remove it from the heat source.
- Slowly Introduce Terpenes: Add your precisely measured terpene blend to the warmed distillate.
- Mix Gently but Thoroughly: Use a low-shear mixer, such as a magnetic stirrer on a low setting, for 2-5 minutes until the mixture is a single, uniform solution. Over-aggressive mixing can introduce oxygen, which accelerates terpene degradation.
Following this controlled method ensures your cannabinoids and terpenes are perfectly homogenized while protecting the full aromatic profile. This disciplined process is the key to consistent, high-quality results.
To make this even more practical, here’s a quick-reference table matching common lab tasks with ideal temperature ranges.
Recommended Temperature Ranges for Formulation Tasks
This table provides practical temperature guidelines. Adhering to these ranges helps preserve terpene integrity while achieving the required viscosity for a premium product.
| Formulation Task | Recommended Temp (°C) | Recommended Temp (°F) | Key Consideration |
|---|---|---|---|
| Mixing Distillate & Terpenes | 65–75°C | 150–167°F | Warm distillate first, then remove from heat before adding terpenes. |
| Filling Cartridges | 70–80°C | 158–176°F | Mixture should be fluid enough to flow easily without being hot enough to degrade terpenes. |
| Short-Term Storage (Pre-Fill) | 20–25°C | 68–77°F | Keep homogenized blends at room temp in a sealed, dark container to prevent degradation. |
| Decarboxylation (Pre-Distillation) | 110–120°C | 230–248°F | Hot enough to activate cannabinoids but low enough to retain some of the hardier terpenes. |
Consider these temperatures as validated starting points. Every extract and terpene blend is slightly different, so use these ranges as a safe zone for dialing in the optimal parameters for your specific product.
How to Store Terpenes to Protect Your Product Quality
Understanding a terpene's boiling point is crucial, but proper storage is equally important for protecting your inventory and ensuring final product quality. The same forces of volatility and vapor pressure are at play when terpenes are sitting on a shelf. Lighter monoterpenes are always seeking to escape, a process accelerated by heat, light, and oxygen.

Best Practices for Terpene Storage
To prevent evaporation and oxidation, a stable, controlled storage environment is necessary. Following these protocols will protect your terpene inventory.
- Keep It Cold: Store all terpene isolates and blends in a cold, dark environment. A dedicated refrigerator set between 2–8°C (35–46°F) is ideal. Cold temperatures dramatically lower vapor pressure, keeping volatile compounds in their liquid state.
- Use the Right Containers: Always use airtight, amber glass bottles or high-grade stainless steel. Amber glass blocks UV light, which can initiate chemical degradation. An airtight seal prevents oxygen ingress and terpene evaporation.
- Minimize Headspace: As you use your terpenes, transfer the remaining liquid to smaller containers. The empty air (headspace) in a partially full bottle provides room for terpenes to evaporate and contains oxygen that can cause oxidation.
- Handle Quickly: When formulating, work efficiently. Open containers, measure what you need, and seal them again immediately. The less time terpenes are exposed to warmer ambient temperatures and open air, the better.
Protecting your terpene profile is a start-to-finish process. The care you take in storing your raw materials is as important as the precision you use during formulation. This discipline guarantees the high-quality experience your brand promises.
By implementing these storage habits, you defend your blends against the gradual loss of volatile top notes like Limonene and Myrcene. This ensures that when you are replicating the flavor of a specific strain, you have the full, vibrant aromatic palette to work with. Proper storage is about consistency; it ensures the last gram of a blend is as potent as the first. Want to dive deeper? You can read also about the many d-Limonene benefits in our guide.
Frequently Asked Questions
Here are answers to common questions we receive from formulators about temperature and terpenes.
At What Temperature Should I Mix Terpenes with Distillate?
The ideal range for mixing terpenes into cannabinoid distillate is 65–75°C (150–167°F). Warm the distillate until it reaches a workable viscosity, then remove it from the heat source before adding your terpenes. Stir them in slowly. This method ensures a homogenous blend while protecting volatile top notes like Pinene and Myrcene. Never add terpenes to distillate while it is still on a hot plate. For a reliable starting point, a good terpene diluent for formulation can make this process much more consistent.
Why Does My Final Product Smell Different Than My Terpene Blend?
If the final product aroma does not match the source terpene blend, it almost always indicates a loss of the most volatile "top notes." Lighter monoterpenes have high vapor pressures and will evaporate quickly when heated or exposed to air for extended periods. To preserve the full profile, maintain mixing temperatures below 75°C, work quickly, and stir gently to avoid agitating the terpenes into the atmosphere.
How Do Boiling Points Affect the Vaping Experience?
A terpene's boiling point determines when it vaporizes during consumption. When a vape device is activated, terpenes with the lowest boiling points vaporize first, delivering the initial burst of flavor. As the heating element remains engaged, terpenes with higher boiling points (mid and base notes) begin to vaporize, causing the flavor to evolve. As a formulator, you can engineer this sensory journey by adjusting the ratio of low-to-high boiling point terpenes.
Can I Use an Old Boiling Points Chart I Found Online?
We strongly advise against it. The internet contains a significant amount of outdated and inaccurate scientific data, particularly concerning cannabis. As demonstrated by the Beta-Caryophyllene example, many widely circulated charts contain critical errors that can lead to failed batches and wasted materials. Rely on data from trusted, lab-verified sources. Using an incorrect boiling point of terpenes chart can cause you to unintentionally degrade valuable compounds and compromise product quality. Our terpene formulation guide is built on current, verified data to help you avoid these costly errors.
At Gold Coast Terpenes, we provide the lab-verified, strain-specific terpene profiles and isolates you need to formulate with confidence. Explore our full collection and elevate your products today.
